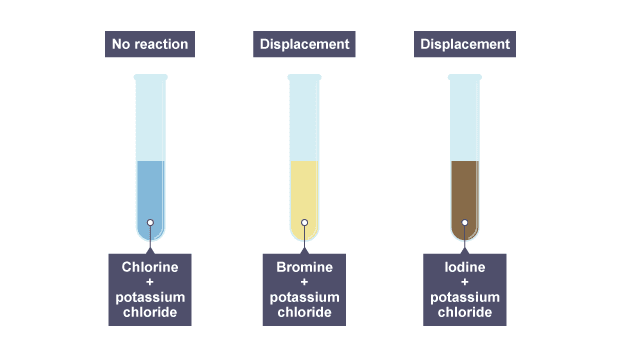 
Halogens are more reactive than hydrogen because, in case of halogens they have 7 valence electrons in their valence shell so they acquire 1 more electron to complete their octet attaining noble / inert gas configuration. Which is more reactive halogen or hydrogen? Its chemical activity can be attributed to its extreme ability to attract electrons (it is the most electronegative element) and to the small size of its atoms. The reaction is faster.įluorine (F), most reactive chemical element and the lightest member of the halogen elements, or Group 17 (Group VIIa) of the periodic table. Has to be warmed and the iron wool heated. Reacts with heated iron wool very quickly. Which is the third most reactive halogen?įluorine is the most reactive element of all in Group 7….Reactivity of halogens. The order of reactivity is chlorine > bromine > iodine. Use the results in the table to deduce an order of reactivity, starting with the most reactive halogen. 
Fluorine is one of the most reactive elements. Halogens can gain an electron by reacting with atoms of other elements. This reactivity is due to high electronegativity and high effective nuclear charge. Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. What is the order of reactivity of halogens?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
